Artificial intelligence chooses best treatment option in breast cancer
To increase the chances of successful surgery for breast cancer, patients sometimes undergo chemotherapy prior to surgery to shrink the tumor. That therapy does not always work, plus it has unpleasant side effects. Researchers can use artificial intelligence to predict for whom pre-operative chemotherapy is useful and for whom it does not. But who leaves such a drastic decision to a computer? In a large international study, Maastricht researchers are translating the technology into practice to increase confidence in artificial intelligence.
Artificial Intelligence
Artificial intelligence is a computer system capable of "learning" from examples. For example, such a system can learn to distinguish between an apple and a pear by analyzing thousands of images of apples and pears and determining which characteristics they differ in. If the system is then shown a new picture of a pear, it will determine which of the two fruits it is based on characteristics such as shape, color and size.
Maastricht University professor Philippe Lambin developed a method for applying artificial intelligence to medical images, such as MRI, CT, or PET scans. This method is called ‘radiomics’. In the case of breast cancer, a computer system learns from MRI scans to distinguish between tumors that shrink due to chemotherapy, and tumors in which chemotherapy has no effect. With MRI scans of new patients, the system then determines whether chemotherapy will be successful, based on the shape and structure of the tumor tissue, for example. If it is not, the patient can be spared chemotherapy, and alternative treatments can be considered more quickly.
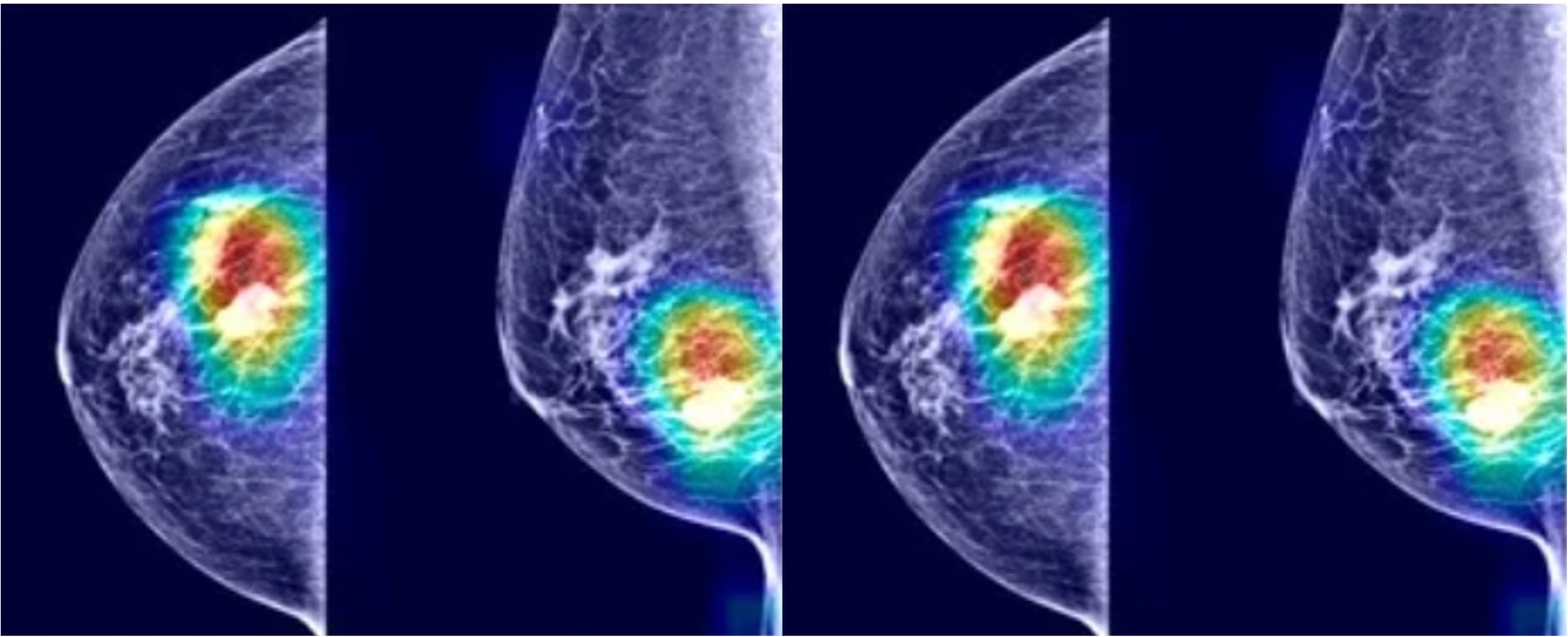
Trust
Although the technology has proven itself, radiomics is not used in healthcare yet. This is not surprising, Lambin believes. "You now get a decision based on artificial intelligence, but the doctor has all kinds of questions with such a decision. Why does the system choose chemotherapy, and not surgery? When does the system think chemotherapy is successful, and when is it not? Doctors will not accept such a decision without proper explanation, without asking questions, or without verifying the decision."
Currently, it is not possible to ask the system questions, but Lambin is changing that. "A kind of dialogue with artificial intelligence," he calls this development. "Suppose the system decides that chemotherapy is the right treatment choice. Then the doctor can, for example, request an MRI scan of a patient in whom chemotherapy was successful, to see the similarities of the scans. Or they can instead study the differences with the MRI scan of a patient in whom chemotherapy did not help. They can also ask the system to estimate the effect of alternative treatment options, or what the consequences would be if the doctor did not follow the recommendation. In this way, doctors can check the decision of artificial intelligence, and determine whether they agree with it. Only when doctors understand how the decision is made will they start using the technology for their patients."
International Research
Translating the scientific technique into a usable method for physicians is part of "RadioVal," a major study in which eight countries from four different continents are collaborating to examine the accuracy, safety and usability of radiomics for breast cancer patients. The study is global for good reason. Lambin: "Radiomics must be able to deal with images from different brands of scanners, with rarer forms of breast cancer, and with forms of breast cancer that are more common in certain continents. The technique must be fair and make a good prediction about the best treatment choice regardless of such differences."
RadioVal is not the only international research building on Lambin's work in radiomics. "Maastricht has now become the European center for radiomics," he says.
The RadioVal project began in September.
Also read
-
Better chances for cancer in the liver
Our liver is a special organ: if you cut away part of it, in most cases a new piece of liver will grow back. If someone has cancer in the liver, the affected part of the liver can be surgically removed. But you can only do this if at least 30% of the liver remains. For many patients whose remaining...

-
-
Diagnosis of skin cancer via quick scan a step closer
Determining whether a suspected spot on the skin is a basal cell carcinoma - the most common form of skin cancer - can be done in a large number of cases with a scan of the skin instead of an invasive biopsy. This has less impact on the patient, is faster and can lead to cost savings in healthcare.
