Improving cancer patient outcomes via evolutionary game theory
Evolutionary game theory can be utilized to improve outcomes in patients with metastatic cancer, according to a new study in the August 9th issue of JAMA Oncology.
The study views cancer therapy as a game between the treating physician and the cancer cells undergoing therapy. The physician “plays” the game by applying a treatment while the cancer cells “play” by evolving new properties that allow them to overcome the toxicity of the drugs administered by the physician. The analysis challenges the decades old standard of treatment for metastatic cancers in which drugs are typically administered continuously at the maximum-tolerated dose (MTD). Instead, the team concluded that treating physicians can exploit their understanding of evolution and their role as the “leader” in the game to apply therapy more strategically to delay or prevent the cancer cells from developing resistance, and ultimately improve patient outcomes.
(text continues below image)
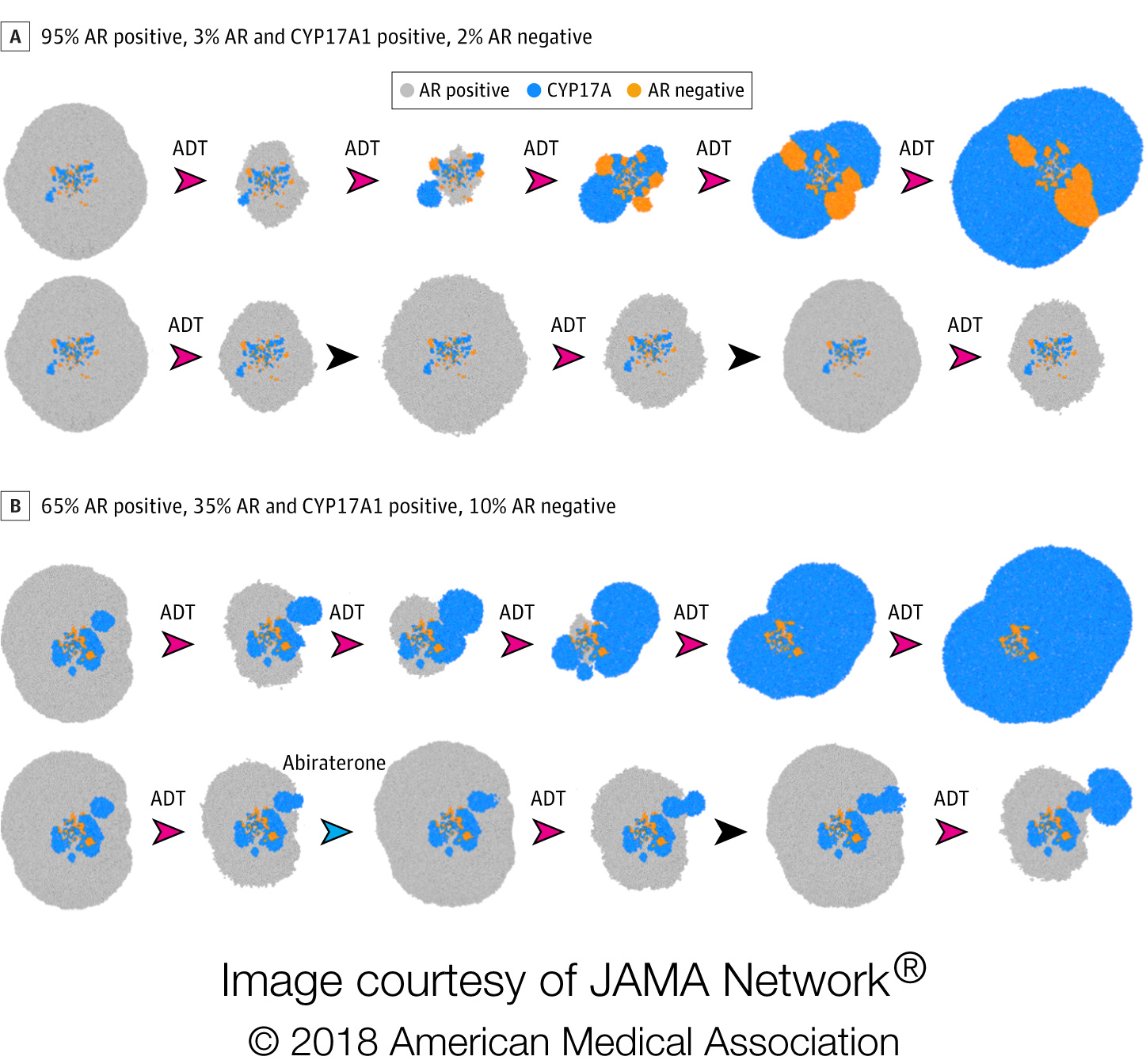
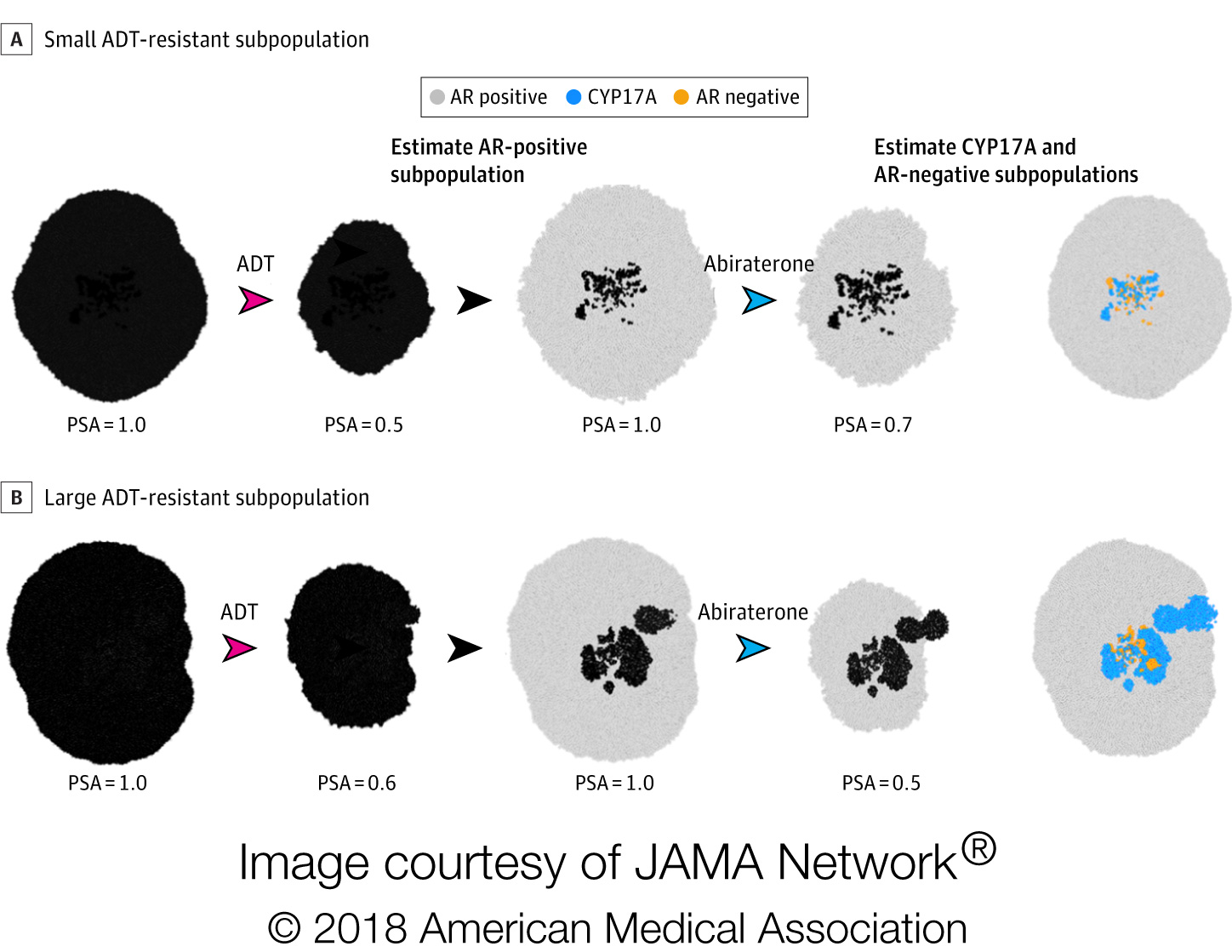
Physician leads
The research team used evolutionary game theory to study the interactions between physicians and cancer cells. In their study, the team demonstrates how the physician can engage the cancer cells in a leader-follower game (often described as a “Stackelberg” game after the German mathematician who extensively investigated the dynamics in the mid-20th century). The physician, who chooses and applies a treatment, is the leader in this game, as he or she has the advantage of “playing first”, while the cancer cells adapt to this treatment and are therefore the followers. Physician’s additional critical advantage is that only the physician is sentient and, therefore, can understand and use the principles of evolution. In contrast, cancer cells, like all evolving populations, can only adapt to current conditions – they cannot think and, therefore, can never anticipate the future or adapt to a treatment that has yet to be applied.
War on cancer
The standard practice for treatment of metastatic cancer, widely accepted for over half a century, does not exploit these advantages. Current therapies typically apply treatments at the maximum-tolerated dose (MTD), which is the largest dose a patient can withstand without excessive toxicity. These doses, often given continually or in repeated cycles, are altered only in the event of unacceptable toxicity or clear evidence of tumour progression. While killing as many cancer cells with the highest possible dose of drug is intuitively appealing in the “war” on cancer, it may be evolutionarily unwise. MTD is likely to accelerate the development of resistance leading to treatment failure. So, even when the initial response to high dose therapy is very favourable in metastatic cancers, resistance typically develops and the cancer recurs.
Improving patient outcomes
“By playing the same strategy repeatedly, current treatments for metastatic cancers greatly increase the speed with which cancer cells can evolve effective counter measures. In fact therapy is usually changed only when the tumour progresses. Ironically, by using this strategy the physician cedes control of the game to the cancer. In effect, the physician becomes the follower. Although standard practice for decades, administering drugs at maximum-tolerated dose until progression represents a poor strategy for most metastatic cancers,” said Gatenby, co-director of Moffitt’s Center of Excellence in Evolutionary Therapy and department chair of Radiology. In order to win the “game”, the physician needs to anticipate how the cancer cells will respond to treatment and what their resistance mechanisms will be. The physician can then use this knowledge to continuously adjust cancer treatments to delay or prevent evolution of resistance in cancer cells and improving patient outcomes.
Adaptive treatment approach
“The current maximum-tolerated dose approach will only be successful if the cancer cell population is made up of similar cells that are unable to adapt and evolve quickly,” added Kateřina Staňková and Joel Brown. “That is rarely what we see for cancers that have widely metastasized. We can and must anticipate, steer and trap the cancer cells’ evolutionary responses to our therapies”.
The research team finds that an adaptive approach will require important but necessary changes to the standard treatment paradigms in the clinic today. “This study shows that physicians can exploit the advantages inherent in the asymmetries of the cancer treatment game, and likely improve outcomes, by adopting more dynamic treatment protocols that integrate eco-evolutionary dynamics and modulate therapy accordingly,” explained Gatenby. Furthermore, while this approach is designed for incurable metastatic cancers, Kateřina Staňková notes that “the full dynamics of Stackelberg games for these applications have not been rigorously explored. As we develop the mathematics in conjunction with cancer therapies, we expect that our analyses will uncover novel game-theoretic, evolutionary strategies that increase the probability of cure in particular in aggressive and heterogeneous cancers.”
About the Department of Data Science and Knowledge Engineering
The Department of Data Science and Knowledge Engineering (DKE) of Maastricht University carries out leading research in artificial intelligence, applied mathematics, operations research, and data science. Key application domains of DKE’s research are in medicine, biology, neuroscience, economics, engineering, and ICT.
About Moffitt Cancer Center
Moffitt is dedicated to one lifesaving mission: to contribute to the prevention and cure of cancer. The Tampa-based facility is one of only 49 US Cancer Institute-designated Comprehensive Cancer Centers, a distinction that recognizes Moffitt’s scientific excellence, multidisciplinary research, and robust training and education.
The study was supported by the European Union's Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement No 690817, the James S. McDonnell Foundation grant, a V Foundation grant and the National Institutes of Health/National Cancer Institute.